Make the invisible visible by a molecular spy
Small molecules such as metabolites, ions and phytohormones function to make a precise decision for plant development. However, with our eyes, we potentially cannot see such molecules; how small molecules are tripping and dancing in plant tissue or organ? This page briefly introduces how we can make the invisible visible.
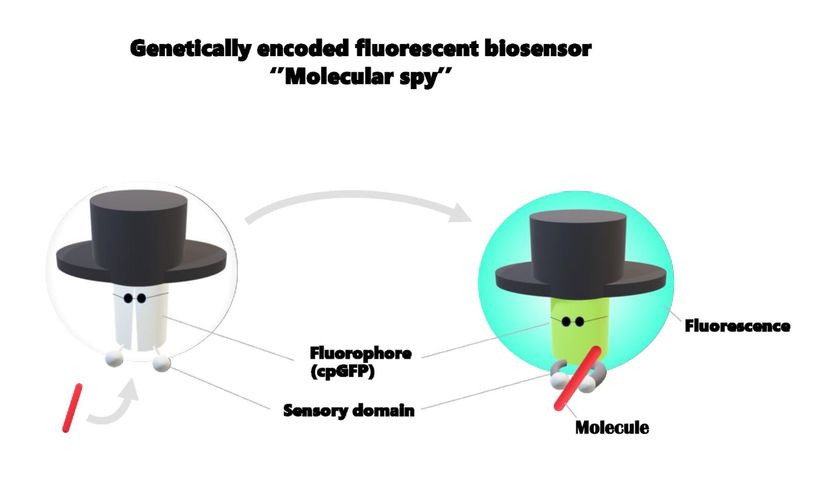
In 1961 Osamu Shimomura unexpectedly discovered the GFP (Green Fluorescence Protein) in the process of aequorin purification from Jellyfish and was awarded the Nobel Prize for Chemistry for his work on this protein. This discovery enabled scientists to colour the various biological phenomena. Because gene for GFP is known, we currently are able to introduce the GFP to the plant cell by a typical transformation method through dipping of agrobacterium. However, GFP cannot detect the small molecules itself, we thus need to put the proper module to sense the analyte of interest, so called sensory domain.
Furthermore, to detect the molecular abundance using GFP, an intensity change of florescence is needed upon sense the analyte by the sensory domain. One of the known technologies to induce this intensity change of fluorescence is “circularly permuted GFP (cpGFP)”. Since the cpGFP protein is folded relatively weak in its three-dimensional structure (conformation), binding of the sensory domain to an analyte induces a conformation change followed by the intensity change of cpGFP fluorescence. Thus, by genetically fusing cpGFP and sensory domain, we are able to make biosensors, and this genetically encoded fluorescent biosensor can tell us the molecular abundance of a specific analyte in the cell.
One of the pioneers of the biosensor field, Roger Tsien, once said that ''our work is often described as building and training molecular spies, molecules that will enter a cell or organism and report back to us what the conditions are''. Thus, genetically encoded sensor is often likened to the ‘’molecular spy’’. But, what does ‘’molecular spy’’ mean?
I imagine that a real spy needs to infiltrate the enemy’s defenses and to report back the valuable information. As a fascinating molecular spy, a biosensor thus needs for the large fluorescence change before and after sensing the analyte of interest, so called ‘’sensitivity’’. High sensitivity results in robust imaging with small noise. The calcium biosensor for example, possesses a high sensitivity shows a robust imaging to visualize the calcium diffusion upon leave wounding and for example as a response to touching in the case of the Venus Flytrap.
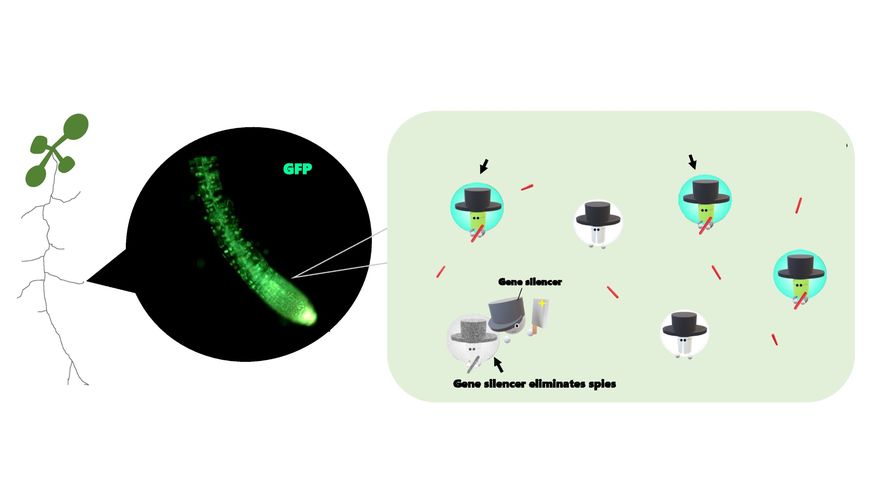
On the other hand, what is the enemy of biosensor within a plant? It seems that “gene silencer” look out for molecular spies to eliminate them. Until now, several papers pointed out that a high abundance of the sensor leads to gene-silencing effect. Therefore, one strategy is to keep the abundance of the biosensor at a minimum in the plant. Another possibility is to use gene silencing mutants, so that we might be able to ignore the gene silencing effect.
In my CEPLAS project, I’m aiming to engineer the fascinating molecular spy which possess the high sensitivity and high accuracy by applying the “Matryoshka technology”. In this technology several fluorescent proteins are used that are genetically encoded in only one section, the concept is reminiscent of the nested Russian “Matryoshka” dolls. These fascinating sensors potentially have good properties to address the molecular transport mechanism during the plant development. Good sensors would be able to provide us with totally new unexpected insights into the molecular mechanisms of the plant.
Planter’s Punch
Under the heading Planter’s Punch we present each month one special aspect of the CEPLAS research programme. All contributions are prepared by our young researchers.
Further Reading
Isoda R, Yoshinari A, Ishikawa Y, Sadoine M, Simon R, Frommer WB, Nakamura M (2020) Sensors for the quantification, localization and analysis of the dynamics of plant hormones. Plant J.
About the Author

Yuuma Ishikawa is postdoctoral researcher at the Institute for Molecular Physiology, Heinrich Heine University Düsseldorf and joined CEPLAS in October 2020. He graduated at the Saitama University in Japan majoring in the Course of Science and Engineering and conferred the degree of Doctor of Engineering. Before he joined CEPLAS, he learned at INRA in France and joined the Frommer group at the Nagoya University in Japan.